 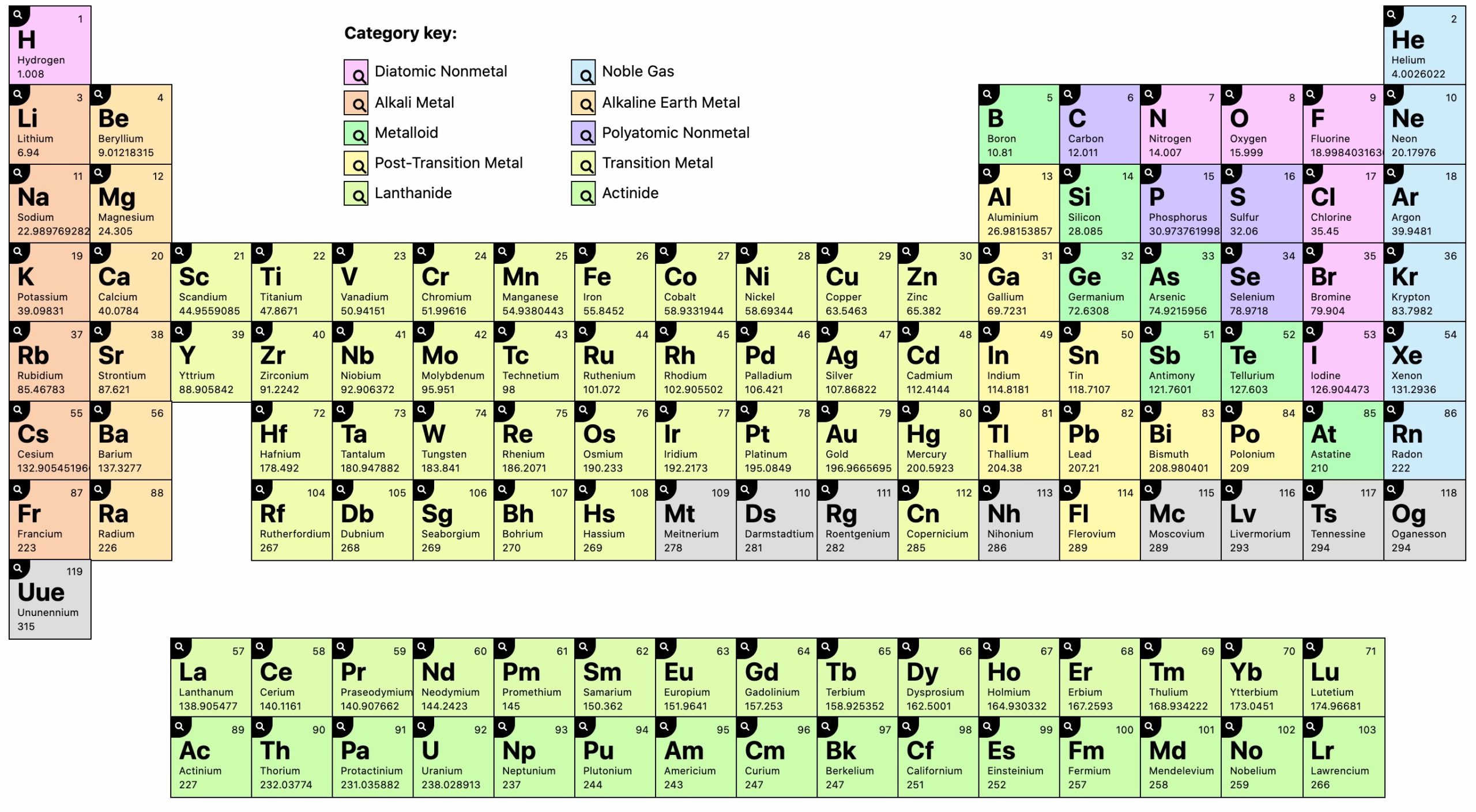
Ruthenium was recognized as a new element hy G.W. The symbols listed below provide examples of the presently accepted form of chemical notation. The number of atoms of an element in a molecule is represented by a subscript written after the symbol thus Au (the first two letters of aurum, the Latin name for gold) represents an atom of gold Cu (the first two letters of cuprum, the Latin name for copper), an atom of copper and C (the first letter of carbon), an atom of carbon O represents an atom of oxygen and 02, a molecule of oxygen. In this notation, the elements are represented by one or two letters, many of which are drawn from the elements Latin or Greek names. Īn internationally accepted chemical notation makes use of symbols to represent elements and compounds, and advises on naming chemical compounds. The symbols Ag and Au for silver and gold come from the Latin names for these elements argentum. The name copper and the symbol Cu are derived from the Latin cuprum, after the island of Cyprus, where the Romans first obtained copper metal. These eleven include seven common metals known to the ancients. Eleven of the elements have names derived from the capitalized first letter of the Latin name of the element and, if necessary, by a second letter (uncapitalized). The symbols are adopted by international agreement among chemists. Thus the element carbon is symbolized by the letter C, the element neon by the letters Ne. Įach element has been named and, for convenience, has been given a nickname-a shorthand symbol of one or two letters. The two elements copper and mercury, which were known in ancient times, have the symbols Cu (cuprum) and Hg (hydrargyrum). :max_bytes(150000):strip_icc()/Elements-58f7944e5f9b581d59396ae7.jpg)
Sometimes the symbol comes from the Latin name of the element or one of its compounds. Thus the symbol for carbon is C that for aluminum is Al. This consists of one or two letters, usually derived from the name of the element. In chemistry, an element is identified by its symbol. Hafnium was named after Hafma, the Latin name for Copenhagen where the discovery was made. 
Moseley s technique of identification was used by means of the x-ray spectra of several 2ircon concentrates and lines at the positions and with the relative intensities postulated by Bohr were found (1). The Bohr atomic theory was the basis for postulating that element 72 should be tetravalent rather than a trivalent member of the rare-earth series. Whereas zirconium was discovered in 1789 and titanium in 1790, it was not until 1923 that hafnium was positively identified. Typically, a symbol is the truncated name of the element or the truncated Latin name of the element. The atomic symbol is one or two letters chosen to represent an element ("H" for "hydrogen," etc.). On the basis of the Bohr theory, the new element was expected to be associated with zirconium. von Hevesey), Hafnium was thought to be present in various minerals and concentrations. Hafinia, Latin name for Copenhagen) Many years before its discovery in 1932 (credited to D. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
